 |
| WILLIAM B. KIOSSES, PHD, THE SCRIPPS RESEARCH INSTITUTE |
Could life have evolved differently? A germ with “unnatural” DNA letters suggests the answer is yes.
E. coli bacteria with an expanded genetic code could help manufacture new drugs.
Every living thing on Earth stores the instructions for life as DNA, using the four genetic bases A, G, C, and T.
All except one, that is.
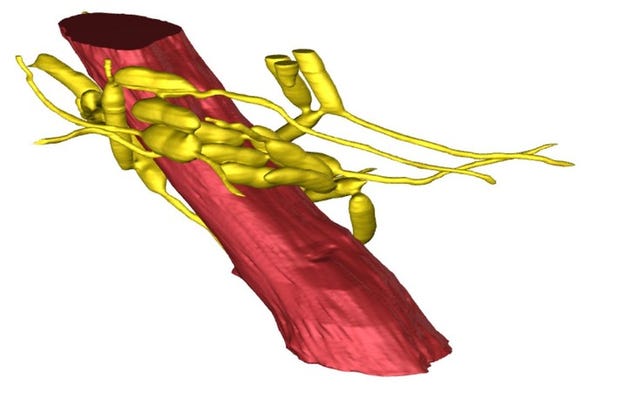
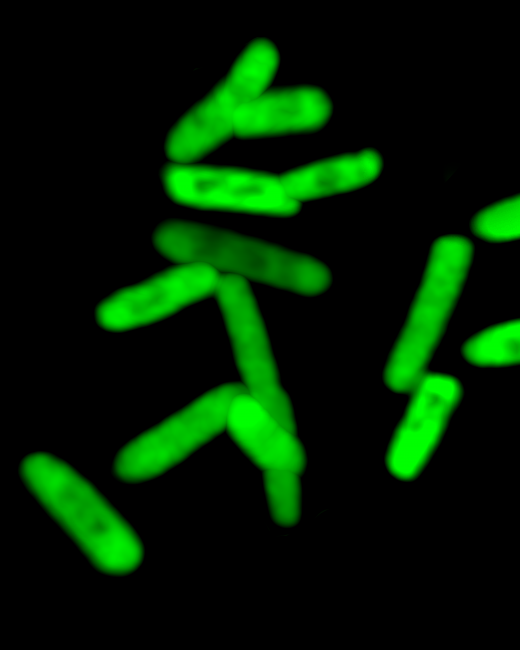
In the San Diego laboratory of Floyd Romesberg—and at a startup he founded—grow bacteria with an expanded genetic code. They have two more letters, an “unnatural” pair he calls X and Y.
Romesberg, head of a laboratory at the Scripps Research Institute, first amended the genes of the bacterium E. Coli to harbor the new DNA components in 2014. Now, for the first time, the germs are using their expanded code to manufacture proteins with equally unusual components.
“We wanted to prove the concept that every step of information storage and retrieval could be mediated by an unnatural base pair,” he says. “It’s not a curiosity anymore.”
The bacterium is termed a “semi-synthetic” organism, since while it harbors an expanded alphabet, the rest of the cell hasn’t been changed. Even so, Peter Carr, a biological engineer at MIT’s Lincoln Laboratory, says it suggests that scientists are only beginning to learn how far life can be redesigned, a concept known as synthetic biology.
“We don’t know what the ultimate limits are on our ability to engineer living systems, and this paper helps show we’re not limited to four bases,” he says. “I think it’s pretty impressive.”
Humankind has been disappointed in the quest to find life on Mars or Jupiter. Yet the alien germs growing in San Diego already hint that our Earth biology isn’t the only one possible. “It suggests that if life did evolve elsewhere, it might have done so using very different molecules or different forces,” says Romesberg. “Life as we know it is may not be the only solution, and may not be the best one.”
Romesberg’s efforts to lay a genetic cuckoo’s egg inside bacteria started 15 years ago. After creating a candidate pair of new genetic letters, the first step was to add them to a bacterium’s genome and show it could use them to store information. That is, could the organism abide by the unnatural DNA and also copy it faithfully as it divided?
The answer, his lab showed in 2014, was yes. But early versions of the bacteria were none too healthy. They died or got rid of the extra letters in their DNA, which are stored in a mini-chromosome called a plasmid. In Romesberg’s words, his creations “lacked the fortitude of real life.”
By this year, the team had devised a more stable bacterium. But it wasn’t enough to endow the germ with a partly alien code—it needed to use that code to make a partly alien protein. That’s what Romesberg’s team, reporting today in the journal Nature, says it has done.
Using the extra letters, they instructed bacteria to manufacture a glowing green protein that has in it a single unnatural amino acid. “We stored information, and now we retrieved it. The next thing is to use it. We are going to do things no one else can,” says Romesberg.
The practical payoff of an organism with a bigger genetic alphabet is that it has a bigger vocabulary—it can assemble proteins with components not normally found in nature. That could solve some tricky problems in medicinal chemistry, which is the art of shaping molecules so they do exactly what’s wanted in the body, and nothing that isn’t.
Pursuing such aims is a startup Romesberg founded, named Synthorx. It has raised $16 million so far and hopes to turn the science into new drugs. One project aims to make a new version of interleukin-2, an anticancer drug with some nasty side effects. Maybe the semi-synthetic germs could fix that by swapping in some unusual components at key points. “This company needs to get out of the lab and into the clinic,” says its newly installed CEO, Laura Shawver.
Carr says an expanded genetic code could have implications beyond providing a shortcut for programming new properties into proteins. He also thinks the new letters might be used to hide information in ways other biologists couldn’t easily see. That could be useful in concealing intellectual property or, perhaps, to disguise a bioweapon.
| Synthorx Inc 2015 |
Credit: William B. Kiosses, PhD, The Scripps Research Institute
ORIGINAL: Technology Review
November 29, 2017
William B. Kiosses, PhD, The Scripps Research Institute